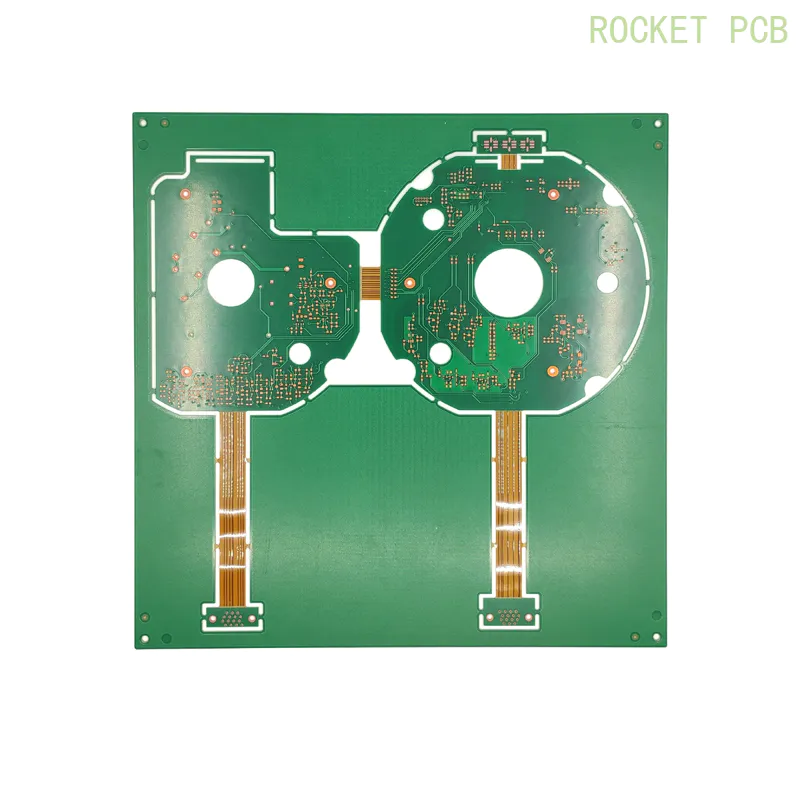
an assay system for point-of-care diagnosis of tuberculosis using commercially manufactured pcb technology
by:Rocket PCB
2019-09-28
Rapid advances in clinical techniques, detection sensitivity, and analytical throughput have greatly expanded our knowledge of prognosis and diagnostic biomarkers for many common infectious diseases such as tuberculosis (TB).
A large number of TB diagnostic methods have been tried in the past decadeof-Care (PoC)
Make use of great changes in technology and materials.
In this work, we describe an electronicbased Enzyme-
Immune adsorption test (eELISA), using a Lab-on-a-
Printed circuit board (LoPCB)
Diagnosis of tuberculosis based on cell factor detection.
The test depends on the Electrochemistry (amperometric)
Analysis, including high
Precision bio-instrument boards and AMP sensors specially produced using standard PCB manufacturing processes.
The use of standard Au and Ag electrodes and customized low
Power supply, multi-channel, portable data
Acquisition system.
We show high
The performance analysis chemistry is performed directly on the micro-fluid volume on the Au pad on the PCB surface, which improves the detection limit (~10u2009pg/mL)
A standard ELISA method.
The analysis was also implemented in plasma, showing the utility of the system in medical applications.
The work is towards building a low
High cost, portable
Precision diagnostic and monitoring technology combined with appropriate PCB
The complete LoPCB platform will be provided based on the micro-flow control network.
The concept of PoC Labs is not new, but it is still an interesting and challenging aspect of clinical diagnosis.
The benefits of PoC labs are multifaceted, and in addition to direct PoC applications, it provides a convenient means to analyze biomarkers in populations far away from hospitals or clinical laboratories.
Moreover, with the continuous expansion of the Internet of Things (IoT)
The PoC Platform can easily connect to the core lab and request any required data or processing support.
Over the past decade, rapid diagnosis of TB in both developed and developing countries has benefited from costs-
Effectiveness of various POC platforms.
However, TB remains one of the world\'s most deadly diseases, and one out of 3 people in the world are affected.
Global, 2014.
6 million people were infected, resulting in 1. 5 million TB-related deaths.
For PoC devices, the obvious route is to migrate standard biochemistry analysis from laboratory or \"analysis tracking\" to PoC diagnostic tools.
However, the large-scale application of PoC technology has been hindered.
The current analysis scheme usually requires chroma detection, so heavy spectral equipment is required, and complex microfluids and pumps need to be customized for manufacturing.
Versatility is also a problem;
For many different analyses, it is clear that many different systems are not suitable for PoC diagnosis, because doctors must have a high degree of confidence in their tools, which is obtained through repeated applications.
There are many reasons why PoC diagnosis has not yet become mainstream in developed economies.
The most important thing is to transfer complex technologies such as pumps, fluids, and complex problems with the transfer of detection units and electrodes to the commercial manufacturing process.
To solve this problem, our philosophy is to develop highly sensitive analytical techniques using specialized commercial PCB manufacturing processes.
Our industrial partner Newbury Electronics manufactures a prototype PCB that includes an entirely embedded micro-fluid structure and an Au/Ag AMP electrode on the surface of the PCB.
This commercially manufactured PCB method allows the production of fully functional amps PCB-
A few pennies per sensor (
Electronic equipment and chemical reagents are not included).
In addition, our design and technology allow any analysis that fits the system on a standard PCB unit by switching reagents.
In this publication, we intend to demonstrate the transformation of electro-chemical analysis technology to a commercial manufacturing surface.
We investigate in detail the integration points needed to develop a working biometric system and work with existing manufacturing technologies to move towards fully working equipment.
We demonstrate a brand new PCB design that improves the sensitivity compared to the commercial hue and the open ampere measurement system, and analyze the pre-
Special reference is made to the calibration performance of clinical diagnosis.
The analysis we implement is a commercially available resistance
Interferon gamma (IFN)
ELISA of R u2009 D scale. IFN is a pro-
Inflammatory cytokines that play a central role in innate and acquired immunity.
Analysis of interferon gamma release in clinical laboratories (IGRA’s)
Routine use in high-risk populations to diagnose potential TB infections (LTBI).
These business analyses are based on T-
Cell response to re in TB patients
Stimulated with TB-specific antigen (CFP-10, ESAT-6 and TB7. 7)
By producing interferon.
Produce T-interferon
Remission of cell ELISAspot (T-
Oxford Center for Immunization technology)
Or interferon secretion detected by ELISA (QuantiFERON-TB Gold In-Tube, cell).
However, IGRA is very time consuming and takes 12 hours in advance
Blood was incubated with TB antigen before blood separation.
In addition, the quantitative test sample is usually a batch test for cost
Effectiveness, increasing the processing time to one week or more.
Previous research work published by our group uses PCB to detail early results
Based on analytical techniques for detecting interferon using commercial potetistat and providing comparison results of other color and AMP efforts.
We now demonstrate that the analytical sensitivity is improved compared to the previously published systems and standard color methods.
In addition, we introduce and apply our own customization
Data acquisition electronic equipment for PCBsensors’ read-out.
In addition to providing separate analysis and separate sensor performance data, we also describe sensor performance and identify measurement protocols related to automated PoC equipment.
Independent characteristic surface plasma resonance is detected. SPR)
, All the measurements are made using custom-
Portable electronic products for home use
We also present the analytical function in the plasma matrix to reflect the application that is ultimately expected.
Our commercial PCB manufacturing technology will lead us to develop a light-weight, low-cost product.
Cost ampere detection device, manufactured using standard commercial processes, including embedded microfluids and multiple
Channel ampere detection.
These analyses are based on the electrochemical detection of 3,3\', 5,5 \'-
4-methyl benzidineTMB)
A common color reporting reagent in standard ELISAs.
We demonstrate the analytical component ligand binding of the standard SPR, allowing comparison with other common analytical systems, and evaluating the binding of captured antibodies (
Single chain antibody modified by cystine)
By connecting with the sulfur bond on the Au surface.
We show the TMB substrate conversion dependent amp signal boost with linear correlation, and we show the complete analytical performance in water and plasma substrates.
This PCB technology is simple and diagnostic analysis of many industry standards can be adapted to this analysis platform.
A large number of TB diagnostic methods have been tried in the past decadeof-Care (PoC)
Make use of great changes in technology and materials.
In this work, we describe an electronicbased Enzyme-
Immune adsorption test (eELISA), using a Lab-on-a-
Printed circuit board (LoPCB)
Diagnosis of tuberculosis based on cell factor detection.
The test depends on the Electrochemistry (amperometric)
Analysis, including high
Precision bio-instrument boards and AMP sensors specially produced using standard PCB manufacturing processes.
The use of standard Au and Ag electrodes and customized low
Power supply, multi-channel, portable data
Acquisition system.
We show high
The performance analysis chemistry is performed directly on the micro-fluid volume on the Au pad on the PCB surface, which improves the detection limit (~10u2009pg/mL)
A standard ELISA method.
The analysis was also implemented in plasma, showing the utility of the system in medical applications.
The work is towards building a low
High cost, portable
Precision diagnostic and monitoring technology combined with appropriate PCB
The complete LoPCB platform will be provided based on the micro-flow control network.
The concept of PoC Labs is not new, but it is still an interesting and challenging aspect of clinical diagnosis.
The benefits of PoC labs are multifaceted, and in addition to direct PoC applications, it provides a convenient means to analyze biomarkers in populations far away from hospitals or clinical laboratories.
Moreover, with the continuous expansion of the Internet of Things (IoT)
The PoC Platform can easily connect to the core lab and request any required data or processing support.
Over the past decade, rapid diagnosis of TB in both developed and developing countries has benefited from costs-
Effectiveness of various POC platforms.
However, TB remains one of the world\'s most deadly diseases, and one out of 3 people in the world are affected.
Global, 2014.
6 million people were infected, resulting in 1. 5 million TB-related deaths.
For PoC devices, the obvious route is to migrate standard biochemistry analysis from laboratory or \"analysis tracking\" to PoC diagnostic tools.
However, the large-scale application of PoC technology has been hindered.
The current analysis scheme usually requires chroma detection, so heavy spectral equipment is required, and complex microfluids and pumps need to be customized for manufacturing.
Versatility is also a problem;
For many different analyses, it is clear that many different systems are not suitable for PoC diagnosis, because doctors must have a high degree of confidence in their tools, which is obtained through repeated applications.
There are many reasons why PoC diagnosis has not yet become mainstream in developed economies.
The most important thing is to transfer complex technologies such as pumps, fluids, and complex problems with the transfer of detection units and electrodes to the commercial manufacturing process.
To solve this problem, our philosophy is to develop highly sensitive analytical techniques using specialized commercial PCB manufacturing processes.
Our industrial partner Newbury Electronics manufactures a prototype PCB that includes an entirely embedded micro-fluid structure and an Au/Ag AMP electrode on the surface of the PCB.
This commercially manufactured PCB method allows the production of fully functional amps PCB-
A few pennies per sensor (
Electronic equipment and chemical reagents are not included).
In addition, our design and technology allow any analysis that fits the system on a standard PCB unit by switching reagents.
In this publication, we intend to demonstrate the transformation of electro-chemical analysis technology to a commercial manufacturing surface.
We investigate in detail the integration points needed to develop a working biometric system and work with existing manufacturing technologies to move towards fully working equipment.
We demonstrate a brand new PCB design that improves the sensitivity compared to the commercial hue and the open ampere measurement system, and analyze the pre-
Special reference is made to the calibration performance of clinical diagnosis.
The analysis we implement is a commercially available resistance
Interferon gamma (IFN)
ELISA of R u2009 D scale. IFN is a pro-
Inflammatory cytokines that play a central role in innate and acquired immunity.
Analysis of interferon gamma release in clinical laboratories (IGRA’s)
Routine use in high-risk populations to diagnose potential TB infections (LTBI).
These business analyses are based on T-
Cell response to re in TB patients
Stimulated with TB-specific antigen (CFP-10, ESAT-6 and TB7. 7)
By producing interferon.
Produce T-interferon
Remission of cell ELISAspot (T-
Oxford Center for Immunization technology)
Or interferon secretion detected by ELISA (QuantiFERON-TB Gold In-Tube, cell).
However, IGRA is very time consuming and takes 12 hours in advance
Blood was incubated with TB antigen before blood separation.
In addition, the quantitative test sample is usually a batch test for cost
Effectiveness, increasing the processing time to one week or more.
Previous research work published by our group uses PCB to detail early results
Based on analytical techniques for detecting interferon using commercial potetistat and providing comparison results of other color and AMP efforts.
We now demonstrate that the analytical sensitivity is improved compared to the previously published systems and standard color methods.
In addition, we introduce and apply our own customization
Data acquisition electronic equipment for PCBsensors’ read-out.
In addition to providing separate analysis and separate sensor performance data, we also describe sensor performance and identify measurement protocols related to automated PoC equipment.
Independent characteristic surface plasma resonance is detected. SPR)
, All the measurements are made using custom-
Portable electronic products for home use
We also present the analytical function in the plasma matrix to reflect the application that is ultimately expected.
Our commercial PCB manufacturing technology will lead us to develop a light-weight, low-cost product.
Cost ampere detection device, manufactured using standard commercial processes, including embedded microfluids and multiple
Channel ampere detection.
These analyses are based on the electrochemical detection of 3,3\', 5,5 \'-
4-methyl benzidineTMB)
A common color reporting reagent in standard ELISAs.
We demonstrate the analytical component ligand binding of the standard SPR, allowing comparison with other common analytical systems, and evaluating the binding of captured antibodies (
Single chain antibody modified by cystine)
By connecting with the sulfur bond on the Au surface.
We show the TMB substrate conversion dependent amp signal boost with linear correlation, and we show the complete analytical performance in water and plasma substrates.
This PCB technology is simple and diagnostic analysis of many industry standards can be adapted to this analysis platform.
Custom message